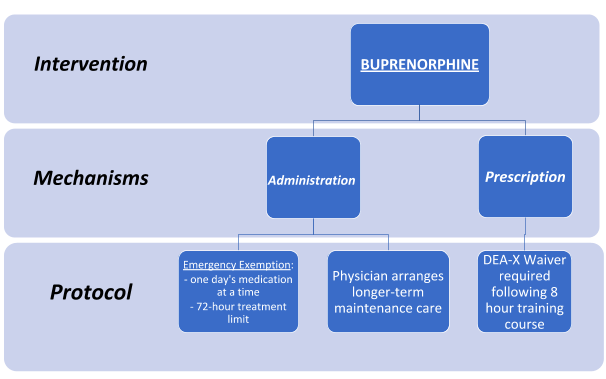
In contrast to methadone for maintenance treatment, buprenorphine enables ED physicians to begin OUD medication-assisted therapy immediately in the emergency department upon presentation. Two routes for its clinical deployment are via administration and prescription:
Administration
Under emergency circumstances, an emergency physician reserves the right to exercise the nonrenewable, non-extendable “three-day rule,” an exemption to the federal mandate requiring a separate DEA-X Waiver to employ narcotic drugs in treating acute opioid withdrawal. In this event, DEA-X Waiver protocol legally allows a provider to administer up to 72 hours of buprenorphine treatment at his or her professional discretion. However, treatment is capped at one day’s limit at a time, thereby requiring multiple visits to continue care within the same ED. This averts the potential for abuse or adverse consequences during the detoxification process. Furthermore, the patient must be referred to longer-term care following treatment.
Prescription
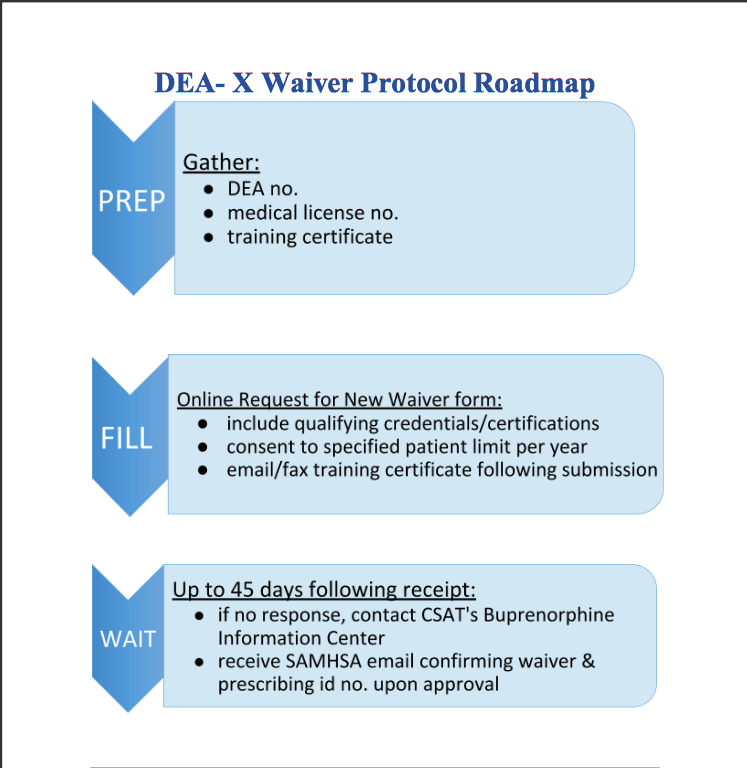
Implementing buprenorphine maintenance therapy requires that physicians obtain a DEA-X Waiver. To do so, practitioners must first register for and then successfully complete a SAMHSA-approved eight-hour training course. Current statistics indicate that only 1% of all emergency medicine physicians have this waiver to prescribe buprenorphine, implying missed opportunities to successfully treat OUD immediately in the ER.
Both mechanisms require a continuity of care that incorporates long-term management within primary care or specialty care environments.