In 2019 and 2020, COVID-19 ravaged worldwide and the World Health Organization (WHO) made various suggestions to decrease the spread of the disease. Many hospitals and clinics limited patient access, and some even closed down temporarily. COVID affected many both directly and indirectly, including those struggling with Opioid Use Disorder. Deaths due to opioid use surged nationwide during the COVID-19 pandemic. Patients who needed to have regular visits to manage their illness found their treatments to be disrupted. Social distancing mandates forced many struggling with addiction to become isolated, increasing the likelihood for substances to be abused. In fact, one study showed that there was a 25% increase in deaths between August 2018- August 2019 and August 2019 – August 2020. (CDC reported this number as underrepresented). According to the National Center for Health Statistics (NCHS), this was the greatest jump seen in 20 years. A similar trend occurred in China, as an increase in suicide attempts through drug overdoses during the 2003 Severe Acute Respiratory Syndrome (SARS) epidemic.
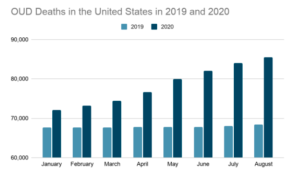
Figure 1- Month by month comparison between 2019 and 2020 for a 12-month ending count of OUD-related deaths in the U.S.
COVID spread quickly in prisons due to overcrowding, and to stifle the spread, many correction facilities released inmates with inadequate support systems. Altogether, major prisons in the U.S. reported having 11% fewer incarcerations during the pandemic in an effort to reduce crowding, and correctional facilities released many homeless individuals struggling with OUD. With an estimate of up to 65% of inmates struggling with some form of substance abuse, overdose is 40 times more likely to occur within weeks after release from prison. This is usually due to a lowered tolerance after abstinence from opioids in jail.
Patients and providers found there were also hurdles to jump in the emergency room. When the pandemic first started in the United States, many OUD treatment facilities announced temporary closings. This, in turn, caused Emergency Physicians to have their hands tied. Those who were able to give patients a “bridge-dose” of buprenorphine did not have treatment facilities to refer patients to. Despite some offices changing to telemedicine after the government allowed it, many individuals did not have ease of access to telemedicine. These issues are not only localized to one area but were faced nationwide.
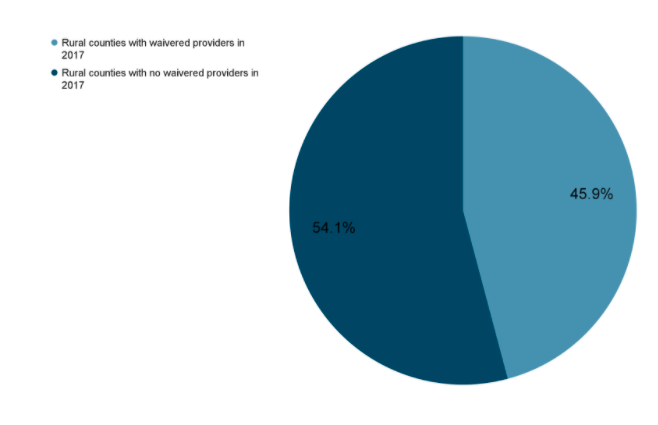
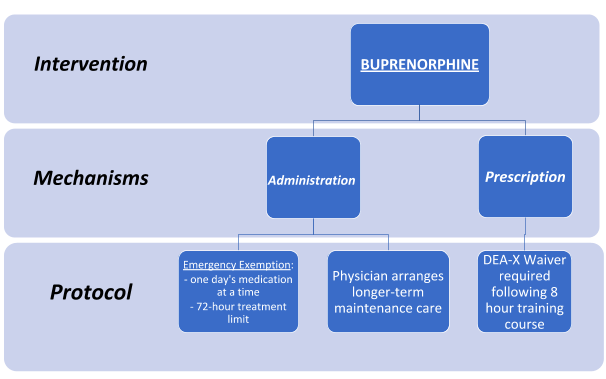
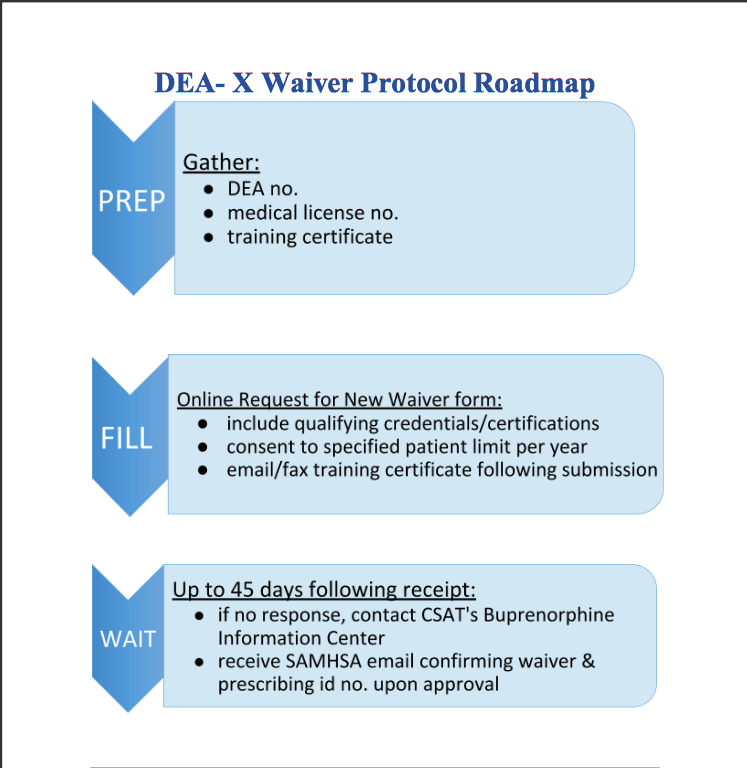
The current administration has discussed some steps, which include: increasing funding and increasing ease of access for medication-assisted training (MAT). One key step providers can make to help reduce the number of people affected by OUD is to obtain a waiver. Having a waiver to prescribe medications such as buprenorphine can help patients start getting the help they need while waiting to go into treatment. This training is available to providers, nurses, and both nursing and medical students. To register for virtual training in your area, please visit <https://getwaivered.com/remote/>.
Biden administration pulls back new guidelines on Prescribing Buprenorphine. (2021, February 05). Retrieved April 15, 2021, from https://www.aamc.org/advocacy-policy/washington-highlights/biden-administration-pulls-back-new-guidelines-prescribing-buprenorphine
Mukherjee, T. I., & El-Bassel, N. (2020). The perfect storm: COVID-19, mass incarceration and the opioid epidemic. International Journal of Drug Policy, 83, 102819. doi:10.1016/j.drugpo.2020.102819
Overdose deaths accelerating during covid-19. (2020, December 18). Retrieved April 15, 2021, from https://www.cdc.gov/media/releases/2020/p1218-overdose-deaths-covid-19.html
Products – DATA Briefs – Number 394 – December 2020. (2020, December 22). Retrieved April 18, 2021, from https://www.cdc.gov/nchs/products/databriefs/db394.htm
Products – vital statistics rapid release – provisional drug overdose data. (2021, March 17). Retrieved April 15, 2021, from https://www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm
Slavova, S., Rock, P., Bush, H. M., Quesinberry, D., & Walsh, S. L. (2020). Signal of INCREASED opioid overdose During COVID-19 from emergency medical services data. Drug and Alcohol Dependence, 214, 108176. doi:10.1016/j.drugalcdep.2020.1081761
The spike in drug overdose deaths during the covid-19 pandemic and policy options to move forward. (n.d.). Retrieved April 15, 2021, from https://www.commonwealthfund.org/blog/2021/spike-drug-overdose-deaths-during-covid-19-pandemic-and-policy-options-move-forward
Wakeman, S., Green, T., & Rich, J. (2020, May 01). An overdose surge will compound the COVID-19 pandemic if urgent action is not taken. Retrieved April 15, 2021, from https://www.nature.com/articles/s41591-020-0898-0
Walter, L. A., & Li, L. (2020). Opioid use disorder in the emergency department amid covid-19. Journal of Addiction Medicine, 14(6). doi:10.1097/adm.0000000000000717